Archive : Article / Volume 2, Issue 2
- Research Article | DOI:
- https://doi.org/10.58489/2836-2217/014
Synthesis And Antibacterial Activity of 6-Bromo-2-(O-Aminophenyl)-3-Amino-Quinazolin-4(3h)-One From 6-Bromo,2-(O-Aminophenyl)-3,1-Benzoxazin-4(3h)-One
1Department of Chemical Science, Ondo State University of Science and Technology, Okitipupa, Ondo State, Nigeria.
Osarumwense Peter Osarodion, Department of Chemical Science, Ondo State University of Science and Technology, Okitipupa, Ondo State, Nigeria.
Osarumwense Peter Osarodion,Synthesis And Antibacterial Activity of 6-Bromo-2-(O-Aminophenyl)-3- Amino-Quinazolin-4(3h)-One From 6-Bromo,2-(O-Aminophenyl)-3,1-Benzoxazin-4(3h)-One. Clin. Case. Rep. Trials. Vol.2,Iss(2). (2023) DOI: 10.58489/2836-2217/014.
© 2023 Osarumwense Peter Osarodion, this is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Received Date: 05-04-2023
- Accepted Date: 13-04-2023
- Published Date: 19-04-2023
Antibacterial activity; Quinazolinone derivatives; 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)- one; 6-bromo, 2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one.
Abstract
Quinazolinone derivatives reveal various medicinal properties such as analgesic, anti-inflammatory and anticancer activities, as well as antimicrobial activity. These heterocycles are valuable intermediates in organic synthesis.Methods/Experimental: The compound, 6-bromo,2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one (1) was synthesized by dissolving 5-bromo anthranillic acid in 100 ml of pyridine. To this reaction mixture o-amino benzoyl chloride stirring at room temperature for 30 minutes this was refluxed with 75 mL of hydrazine hydrate for 3 hrs at 120-1300C. the reaction mixture was allowed to cool to room temperature to give 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)-one (2). These Compounds were evaluated for their bacterialrial activity (against some gram positive and gram negative microorganism) and antifungal activity (against Candida albicans). Study Design: This study was experimentally design and the antibacterial activity was evaluated against some microorganism, Staphylococcus aureus, Bacillus species, Aspergillus Species, Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumonia, and candida albicans Result: The compounds exhibited significant antibacterial activity with a zone of inhibition in the range of 10 – 16mm in comparison to control. Conclusions: From our findings, the compounds synthesized have higher antibacterial activities as compared to Ciprofloxicin (CPX) and Ketonaxol (PEF) standard antibacterial drugs.
Introduction
Quinazoline derivatives, which belong to the N-containing heterocyclic compounds, have caused universal concerns due to their widely and distinct biopharmaceutical activities. Researchers have already determined many therapeutic activities of quinazoline derivatives, including anti-cancer [1–4], anti-inflammation [5, 6], anti-bacterial [7–10], analgesia [5, 9], anti-virus [11], anti-cytotoxin [12], anti-spasm [9, 13], anti-tuberculosis [14], anti-oxidation [15], anti-malarial [16], anti-hypertension [17], anti-obesity [18], anti-psychotic [19], anti-diabetes [20], etc.
Quinazolinone derivatives have been attracting growing attention from medicinal and agricultural chemists, owing to their diverse biological activities, such as antibacterial [21–27], antifungal [28–30], antiviral [31,32], antitumor [33,34] and anticonvulsant [35] activities.
Heterocyclic chemistry comprises at least half of all organic chemistry research worldwide. In particular, heterocyclic structures form the basis of many pharmaceutical, agrochemical and veterinary products. Among a wide variety of nitrogen heterocycles that have been explored for developing role in medicinal chemistry and subsequently have emerged as a pharmacophore [36].
This research was aimed at synthesis of 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)-one and 6-bromo,2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one and investigating them for their antibacterial activity and to obtain more precise information about the course of reaction.
Chemistry
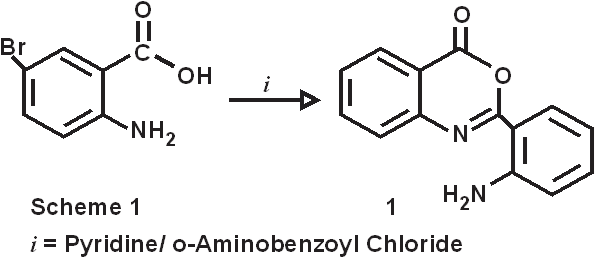
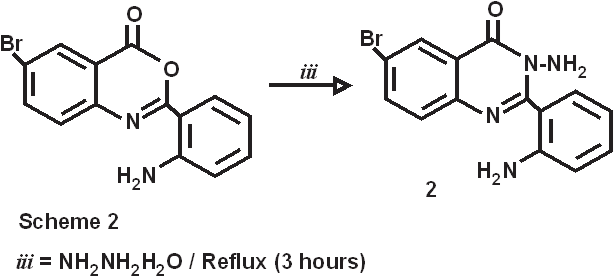
The introduction of 2-amino substituent is a successful strategy to improve the chemical stability of benzoxazinone. Due to the antibacterial and pharmacological activities of 4(3H)-quinazolinone derivatives, 2,3-disubstituted derivative of quinazoline-4-one were synthesized via the interaction of the benzoxazinone derivative with nitrogen nucleophile with the aim of obtaining more pricise information about the course of the reaction and some interesting pharmaceutical compounds. Dissolving 5-bromo anthranillic acid in 100 ml of pyridine in o-amino benzoyl chloride stirring at room temperature for 30 minutes produce the cyclic compound 6-bromo,2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one(1). The reaction of this compound with 75 mL of hydrazine hydrates for 3 hrs at 120-1300C. the reaction mixture was allowed to cool to room temperature to give 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)-one (2).
Materials and Methods
Experimental
All reagents and solvents were purchased from sigma-Aldrich, in Germany. Melting points were determined on a kofler hot stage apparatus and were uncorrected. IR spectra were recorded on a Buck scientific IR M500 instrument. The 1H and 13C NMR spectra were recorded in DMSO-d6 at 400 MHz with HAZ VOLATILE V2. M Chemical shifts Sare reported in ppm relative to tetramethylsilane. Gas chromatography mass spectra were obtained on a Finingan MAT 44S mass spectrophotometer operating at 70eV. Elemental analysis agreed favourably with the calculated values. Analytical thin layer chromatography (TLC) was used to monitor the reactions.
Elemental Analysis
The compositions of the compounds are summarized in table 1. The C and H contents (both theoretically calculated values and actual values) are indicated.
Synthesis of 6-bromo,2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one (1). 5-bromo anthranillic acid (.16M, 34.72gm) was dissolved in 100 ml of pyridine. To this reaction mixture o-amino benzoyl chloride (.16M, 24.8gm) was added with stirring at room temperature. Stirring continued for 30 mins at the same temperature. This reaction mixture was filtered out and collect the precipitate, which was washed with distilled water and Pet.ether 60/80 to remove the traces of pyridine. The pale creamish crystals obtained were dried at 600C. m.p.-1900C, yield- 75%, Synthesis of 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)-one ( 2). 6-bromo-2-(o-aminophenyl)-3-,1-benzoxazin-4(3H)-one (0.075M, 23.775gm) was refluxed with 75 mL of hydrazine hydrate for 3 hrs at 120-1300C. the reaction mixture was allowed to cool to room temperature. Pale creamish crystals developed were recrystallized from super dry ethanol. m.p.-178-1800C, yield-75%,
Antimicrobial Activities
Determination of zone of inhibition
The microbial growth inhibitory activities of the powdered crude drug obtained were determined by the agar well plate method where the compounds were initially dissolved in distilled water (1:1). Those compounds with activities were later tested at concentrations of 10, 15, 20, 60 mg/mL against clinical isolated Staphylococcus aureus, Bacillus species, Escherichia coli, Aspergillus Species, Klebsiella pneumonia, Pseudomonas Aeuriginosa and Candida albicans using the standard microbiological method. Sterile nutrient and Sabouraud dextrose agar plates were prepared for bacteria and fungi respectively and standardized inoculum of test organisms was spread uniformly.
We used a sterile borer (8 mm) and 100μL of the test concentrations, to bored six wells, standard antibiotic, and the solvent control were added to each well. The plates were left on the table for 1 h for the test solution to diffuse into the medium and then incubated at 37°C for 18-24 h. The resultant zone of inhibitions of microbial growth around the well was measured in mm. The test was performed in triplicate. Standard antibiotics ciprofloxacin (30 mg/mL), and Ketonaxol (50 mg/mL) were tested against bacteria and fungi respectively as the positive control [37].
Determination of MIC
The minimum inhibitory concentration (MIC) values of the powdered crude drug obtained were determined using the agar dilution method. Four different concentrations range of 100 µL of the synthesized compounds were incorporated into their respective molten agar and allowed to set. This was also repeated for ciprofloxacin and itraconazole as positive control and the diluent as a negative control. Each of the standardized test microorganisms was radially streaked onto the prepared plates. The plate was left to stand for 1 h at room temperature, incubated at 37°C for 18-24 h. The MIC was recorded as the lowest concentrations that inhibited the growth of each of the test organisms [37].
Results and Discussion
|
Compound No
|
Solvent
|
Formula M. wt
|
Analysis% Calc/Found
|
|
|
C
|
H
|
|||
|
1
|
Ethanol
|
C14H9BrN202 (396.8)
|
42.40
42.50
|
2.27
2.28
|
|
2
|
Ethanol
|
C14H9BrN40 (424.8)
|
39.50
39.70
|
2.11
2.12
|
Table 1: Characterization AndPhysical Data Of Synthesized Compounds
|
Compound No
|
δ (ppm) Carbon atom number
|
 |
157.14(C-1),160.18(C-2),121.13(C-3),127.19(C-4),
112.62(C-5), 112.31(C-6), 121.11 (C-7), 145.06 (C-8),
24.02 (C-9) 112.64(C-10), 112.41(C-11), 112.22 (C-12),
116.07 (C-13), 112.14(C-14).
|
 |
155.32(C-1), 161.12 (C-2), 121.17(C-3), 127.33 (C-4),
112.11 (C-5), 112.12 (C-6), 122.22 (C-7), 147.14(C –
8),24.12 (C- 9), 112.51 (C-10), 112.31 (C-11), 121.11
(C-12),116.09(C – 13), 112.21(C – 14).
|
Table 2: 13C-NMR Of Synthesized Compounds
|
Compound No
|
δ (ppm)
|
 |
5.64 (d, 2H of –NH2), 7.18-7.25 (dd, 1H of-ArH), 7.43-
7.86 (m, 5H of –ArH), 6.71 (d, 1H of -ArH)
|
 |
7.72-7.80 (m, 5H of –ArH), 5.65 (s, 2H of NH2), 6.18 (s,
2H of –NH2).
|
Table 3: 1H-NMR Of Synthesized Compounds
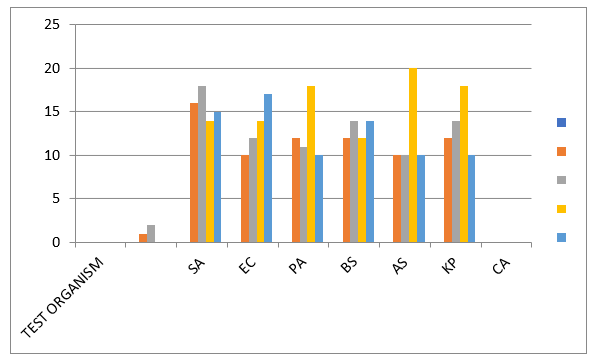
Figure 1: The effect of the Synthesized Compounds and Standard drugs toward studied bacteria. SA = Staphylococcus aureus, BS= Bacillus species, AS = Aspergillus Species, PA = Pseudomonas aeruginosa, EC = Escherichia coli, KP=Klebsiella pneumonia, and CA=candida albicans.
Control drugs- Ciprofloxicin (CPX) for bacteria, Ketonaxol (PEF) for fungus, Compound 1 (1), Compound 2 (2) Significantly different from Ligand at P< 0>
Discussion
The present study reported the synthesis of two derivatives of quinazolinone, 6-bromo,2-(o-aminophenyl)-3,1-benzoxazin-4(3H)-one( 1) and 6-bromo-2-(o-aminophenyl)-3-amino-Quinazolin-4(3H)-one ( 2) . The compounds were investigated for their Antibacterial activity.
Structural elucidations of compounds synthesized were characterized by correct elemental analysis and careful inspections of spectral data. Looking at the 1H NMR spectra of the compounds synthesized, compound 1 displayed a duplet at δ 5.64 which was due to amino, –NH2 group. Other duplet appeared at δ 7.18 and 7.25 attributed to aromatic protons. Also, 1H NMR spectrum of compound 2 showed a characteristic signal at δ 5.65 and 6.18 (singlet) corresponding to the two amino, –NH2 groups. Two singlets appeared at δ7.41 and 7.10 attributed to aromatic protons. Another signal appeared at 5.80 which is attributed to the protons of the amino group. For the IR spectra, compound 1 were characterized by the presence of 3068 υ C-H str. of the aromatic ring, 1698 cm-1 υ C=O str. of the ring, 3365 cm-1 3345 cm-1 υ N-H str. of the ring in the region of the compound. Compound 2 was characterized by presence of υ 3048 cm-1 υ (C-H str. of the aromatic ring), 3361 cm-1 , 3351 cm-1 υ (N-H str. of the ring), 1706 cm-1 υ (C=O str. of the ring), υ 1316 cm-1 region of the compound.
The 13C NMR spectrum of compound 1, revealed signals at δ24.02, attributed to phenyl group, while the aromatic carbon atoms appeared between δ values 112.31 – 160.18 with the carbonyl carbon atom appearing as the highest δ value of 160.18. Similarly, compound 2 showed signals at δ24.12, attributed to phenyl group, while the aromatic carbon atoms appeared between δ values 105.64 - 160.28, with the carbonyl carbon atom appearing as the highest δ value of 160.28.
These compounds synthesized exhibited promising Antibacterial activities. The antibacterial activity of compounds synthesized were determined using the agar well plate method and the results obtained are summarized in Figure 1. Compound 2 showed the highest activity against Staphylococcus aureus, Bacillus species, Escherichia coli, Klebsiella pneumonia compared to the other compound 1. It may be that the substitution of amino group at position three increases the activity. These compounds synthesized have a higher activity against Staphylococcus aureus, than Ciprofloxacin (CPX) and Ketonaxol (PEF), which are standard antibacterial drugs.
Conclusion
The present study has showed that the quinazolinone derivatives 1 and 2 have high antibacterial activity. Compound 2 showed the highest activity against Staphylococcus aureus, Bacillus species, Escherichia coli, Klebsiella pneumonia compared to the other compound 1. It may be that the substitution of amino group at position three increases the activity. These compounds synthesized have a higher activity against Staphylococcus aureus, than Ciprofloxacin (CPX) and Ketonaxol (PEF), which are standard antibacterial drugs. This study has confirmed that the antibacterial analysis shows that the compounds synthesized have high activity against Staphylococcus aureus, Bacillus species, Aspergillus Species, Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumonia, with no activity against Candida albicans. From this result, Compound 2 could be a potential Antibacterial and a tool to pharmaceutical drug delivery.
Declarations
Conflict of interest
The author declares no conflict of interest.
Funding
No fund was obtained during the research.
Author declaration
The author hereby declares that the work presented in this article is original and that any liability for claims relating to the content of this article will be borne by me.
Ethics approval and consent to participate
Ethic approval, consent to participate and the procedure used was approved by the Ethic approval committee of Ondo State University of Science and Technology, Okitipupa, Ondo State, Nigeria.
Acknowledgements
The author acknowledges the assistance of Baba Haruna of the Department of Pharmaceutical Chemistry of Niger Delta University, Wilberforce Island, Yenogoa and Dr. Marvis, in England for running the spectra.
Declaration statement
The author declares there is no conflict of interest.
References
- Chandregowda, Venkateshappa, A. K. Kush, and G. Chandrasekara Reddy. “Synthesis and in vitro antitumor activities of novel 4-anilinoquinazoline derivatives.” European journal of medicinal chemistry 44, no. 7 (2009): 3046-3055.
- Al-Rashood, Sarah T., Ihsan A. Aboldahab, Mahmoud N. Nagi, Laila A. Abouzeid, Alaa AM Abdel-Aziz, Sami G. Abdel-Hamide, Khairia M. Youssef, Abdulrahman M. Al-Obaid, and Hussein I. El-Subbagh. “Synthesis, dihydrofolate reductase inhibition, antitumor testing, and molecular modeling study of some new 4 (3H)-quinazolinone analogs.” Bioorganic & medicinal chemistry 14, no. 24 (2006): 8608-8621.
- Vasdev, Neil, Peter N. Dorff, Andrew R. Gibbs, Erathodiyil Nandanan, Leanne M. Reid, James P. O’Neil, and Henry F. Van- Brocklin. “Synthesis of 6-acrylamido-4-(2-[18F] fluoroanilino) quinazoline: a prospective irreversible EGFR binding probe.” Journal of Labelled Compounds and Radiopharmaceuticals: The Official Journal of the International Isotope Society 48, no. 2 (2005): 109-115.
- Wakeling, Alan E., Simon P. Guy, Jim R. Woodburn, Susan E. Ashton, Brenda J. Curry, Andrew J. Barker, and Keith H. Gibson. “ZD1839 (Iressa) an orally active inhibitor of epidermal growth factor signaling with potential for cancer therapy.” Cancer research 62, no. 20 (2002): 5749-5754.
- Alagarsamy, V., V. Raja Solomon, and K. Dhanabal. “Synthesis and pharmacological evaluation of some 3-phenyl-2-substituted- 3H-quinazolin-4-one as analgesic, anti-inflammatory agents.” Bioorganic & medicinal chemistry 15, no. 1 (2007): 235-241.
- Baba, Atsuo, Noriaki Kawamura, Haruhiko Makino, Yoshikazu Ohta, Shigehisa Taketomi, and Takashi Sohda. “Studies on disease-modifying antirheumatic drugs: synthesis of novel quinoline and quinazoline derivatives and their anti-inflammatory effect.” Journal of medicinal chemistry 39, no. 26 (1996): 5176-5182.
- Rohini, Rondla, P. Muralidhar Reddy, Kanne Shanker, Anren Hu, and Vadde Ravinder. “Antimicrobial study of newly synthesized 6-substituted indolo [1, 2-c] quinazolines.” European journal of medicinal chemistry 45, no. 3 (2010): 1200-1205.
- Antipenko, Lyudmila, Alexander Karpenko, Sergey Kovalenko, Andrew Katsev, Elena Komarovska-Porokhnyavets, Vladimir Novikov, and Aleksey Chekotilo. “Synthesis of new 2-thio-[1, 2, 4] triazolo [1, 5-c] quinazoline derivatives and its antimicrobial activity.” Chemical and Pharmaceutical Bulletin 57, no. 6 (2009): 580-585.
- Gupta, Vivek, Sushil K. Kashaw, Varsha Jatav, and Pradeep Mishra. “Synthesis and antimicrobial activity of some new 3–[5-(4-substituted) phenyl-1, 3, 4-oxadiazole-2yl]-2-styrylquinazoline- 4 (3H)-ones.” Medicinal Chemistry Research 17, no. 2 (2008): 205-211.
- Aly, A. A. “Synthesis of novel quinazoline derivatives as antimicrobial agents.” Chinese Journal of Chemistry 21, no. 3 (2003): 339-346.
- Li H, Huang R, Qiu D, Yang Z, Liu X, Ma J, Ma Z: Synthesis and bioactivity of 4-quinazoline oxime ethers. Prog Nat Sci. 1998, 8: 359-365.
- Chandrika, P. Mani, T. Yakaiah, B. Narsaiah, V. Sridhar, G. Venugopal, J. Venkateshwara Rao, K. Pranay Kumar, U. S. N. Murthy, and A. Raghu Ram Rao. “Synthesis leading to novel 2, 4, 6-trisubstituted quinazoline derivatives, their antibacterial and cytotoxic activity against THP-1, HL-60 and A375 cell lines.” Indian J Chem. B 48 (2009): 840-47.
- Paneersalvam, P., T. Raj, M. P. S. Ishar, B. Singh, V. Sharma, and BAo Rather. “Anticonvulsant activity of Schiff bases of 3-amino-6, 8-dibromo-2-phenyl-quinazolin-4 (3H)-ones.” Indian journal of pharmaceutical sciences 72, no. 3 (2010): 375.
- Nandy, Parijat, M. T. Vishalakshi, and A. R. Bhat. “Synthesis and antitubercular activity of Mannich bases of 2-methyl-3Hquinazolin- 4-ones.” Indian Journal of heterocyclic chemistry 15, no. 3 (2006): 293.
- Saravanan, G. O. V. I. N. D. A. R. A. J., V. E. E. R. A. C. H. A. M. Y. Alagarsamy, and CHINNASAMY RAJARAM Prakash. “Synthesis and evaluation of antioxidant activities of novel quinazoline derivatives.” Int J Pharm Pharm Sci 2, no. 4 (2010): 83-86.
- Lakhan R, Singh OP, Singh-J RL: Studies on 4 (3H)-quinazolinone derivatives as anti-malarials. J Indian Chem Soc. 1987, 64: 316-318.
- Hess, Hans J., Timothy H. Cronin, and Alexander Scriabine. “Antihypertensive 2-amino-4 (3H)-quinazolinones.” Journal of medicinal chemistry 11, no. 1 (1968): 130-136.
- Sasmal, Sanjita, D. Balasubrahmanyam, Hariprasada R. Kanna Reddy, Gade Balaji, Gujjary Srinivas, Srisailam Cheera, Chandrasekhar Abbineni et al. “Design and optimization of quinazoline derivatives as melanin concentrating hormone receptor 1 (MCHR1) antagonists: part 2.” Bioorganic & medicinal chemistry letters 22, no. 9 (2012): 3163-3167.
- Alvarado, Mario, María Barceló, Laura Carro, Christian F. Masaguer, and Enrique Raviña. “Synthesis and biological evaluation of new quinazoline and cinnoline derivatives as potential atypical antipsychotics.” Chemistry & biodiversity 3, no. 1 (2006): 106-117.
- Malamas, Michael S., and Jane Millen. “Quinazolineacetic acids and related analogs as aldose reductase inhibitors.” Journal of medicinal chemistry 34, no. 4 (1991): 1492-1503.
- Sharma, Pratibha, Ashok Kumar, Prerna Kumari, Jitendra Singh, and M. P. Kaushik. “QSAR modeling of synthesized 3-(1, 3-benzothiazol-2-yl) 2-phenyl quinazolin-4 (3 H)-ones as potent antibacterial agents.” Medicinal Chemistry Research 21, no. 7 (2012): 1136-1148.
- Wang, Xiang, Juan Yin, Li Shi, Guoping Zhang, and Baoan Song. “Design, synthesis, and antibacterial activity of novel Schiff base derivatives of quinazolin-4 (3H)-one.” European journal of medicinal chemistry 77 (2014): 65-74.
- Bouley, Renee, Derong Ding, Zhihong Peng, Maria Bastian, Elena Lastochkin, Wei Song, Mark A. Suckow et al. “Structure– activity relationship for the 4 (3 H)-Quinazolinone antibacterials.” Journal of medicinal chemistry 59, no. 10 (2016): 5011- 5021.
- Yang, X., Wang, X., & Wu, M. (2014). Synthesis and biological properties of 3-(2-hydroxyethyl)-2- (phenylamino) quinazolin-4 (3H)-ones. Chinese Journal of Organic Chemistry, 34(5), 1015.
- Veena Vani, Kotla, Garlapati Ramesh, and Chunduri Venkata Rao. “Synthesis of New Triazole and Oxadiazole Derivatives of Quinazolin-4 (3H)-one and Their Antimicrobial Activity.” Journal of Heterocyclic Chemistry 53, no. 3 (2016): 719-726.
- Asadi, Parvin, Ghadamali Khodarahmi, Ali Jahanian-Najafabadi, Lotfollah Saghaie, and Farshid Hassanzadeh. “Biologically active heterocyclic hybrids based on quinazolinone, benzofuran and imidazolium moieties: synthesis, characterization, cytotoxic and antibacterial evaluation.” Chemistry & Biodiversity 14, no. 4 (2017): e1600411.
- Asker, Firyal Weli, Salma Abdul Rudha Abbas, Redha Ibrahim Al-Bayati, and Hanaa Abd Al-Tamemi. “Synthesis and biological evaluation of new quinazolinone derivatives.” European Journal of Chemistry 5, no. 4 (2014): 628-634.
- Prashanth, M. K., and H. D. Revanasiddappa. “Synthesis of some new glutamine linked 2, 3-disubstituted quinazolinone derivatives as potent antimicrobial and antioxidant agents.” Medicinal Chemistry Research 22, no. 6 (2013): 2665-2676.
- Mohamed, Mosaad S., Mohsen M. Kamel, Emad MM Kassem, Nageh Abotaleb, Sherein I. Abd El-moez, and Marwa F. Ahmed. “Novel 6, 8-dibromo-4 (3H) quinazolinone derivatives of anti-bacterial and anti-fungal activities.” European Journal of Medicinal Chemistry 45, no. 8 (2010): 3311-3319.
- Mabkhot, Yahia Nasser, Munirah S. Al-Har, Assem Barakat, Fahad D. Aldawsari, Ali Aldalbahi, and Zaheer Ul-Haq. “Synthesis, anti-microbial and molecular docking studies of quinazolin-4 (3H)-one derivatives.” Molecules 19, no. 7 (2014): 8725-8739.
- Kumar, Krishnan Suresh, Swastika Ganguly, Ravichandran Veerasamy, and Erik De Clercq. “Synthesis, antiviral activity and cytotoxicity evaluation of Schiff bases of some 2-phenyl quinazoline-4 (3) H-ones.” European journal of medicinal chemistry 45, no. 11 (2010): 5474-5479.
- Gao, Xingwen, Xuejian Cai, Kai Yan, Baoan Song, Lili Gao, and Zhuo Chen. “Synthesis and antiviral bioactivities of 2-arylor 2-methyl-3-(substituted-benzalamino)-4 (3 H)-quinazolinone derivatives.” Molecules 12, no. 12 (2007): 2621-2642.
- Wang, S., Gao, M., Tan, G., Ma, H., Zhao, Y., Du Hongyuan, W. Z., ... & Li, X. (2017). Effects of the substituent at C-2 phenyl on the N-/O-alkylation of quinazolin-4 (3H)-one and anti-tumor, antimicrobial activities of some compounds. Chinese Journal of Organic Chemistry, 37(2), 385.
- Mohamed, Menshawy A., Rezk R. Ayyad, Taghreed Z. Shawer, Alaa A-M. Abdel-Aziz, and Adel S. El-Azab. “Synthesis and antitumor evaluation of trimethoxyanilides based on 4 (3H)-quinazolinone scaffolds.” European journal of medicinal chemistry 112 (2016): 106-113.
- Noureldin, Nada A., Hend Kothayer, El-Sayed M. Lashine, Mohamed M. Baraka, Wafaa El-Eraky, and Sally A. El Awdan. “Synthesis, anticonvulsant activity, and SAR study of novel 4-quinazolinone derivatives.” Archiv der Pharmazie 350, no. 2 (2017): 1600332.
- Banu, B. Haseena, K. Bharathi, and K. V. S. R. G. Prasad. “Synthesis, characterization and evaluation of in vitro antioxidant and anti-inflammatory activity of 2-(4-oxo-2-phenylquinazolin-3 (4H)-yl) substituted acetic acids.” IOSR J Pharm 2 (2012): 97- 104.
- Osarumwense, Peter Osarodion, Mary Olire Edema, and Cyril Odianosen Usifoh. “Synthesis and antibacterial activities of quinazolin-4 (3H)-one, 2-methyl-4 (3H)-quinazolinone and 2-phenyl-4 (3H)-quinazolinone.” Int. J. Biol. Pharm. Sci. Arch 1, no. 2 (2021): 077.


